MEMPHIS: Modular Evaluation of immunogenicity using Multi-Platform Human In vitro Systems
Background
Though vaccination is one of the greatest accomplishments of modern medicine, there are still challenges to the production of perfectly effective vaccines. These challenges include weakened response by the vaccinated individual’s immune system as well as time-limited immunization against specific organisms which many mean additional doses of the vaccine are required at a later time to boost initial immunization.
To overcome weak immune response to vaccination, scientist have discovered that specific adjuvant formulations, like lipid nanoparticles, oil-in-water emulations, and aluminum salts, can promote stronger and prolonged immune responses. However, despite the success of adding these adjuvants, the mechanisms underlying their actions are still not fully understood. To identify the best adjuvant for a vaccine, extensive in vitro research on specific cell types, or in vivo research in model organisms, is required. There are also challenges in identifying the best adjuvants due to: (1) the variations among different immune cells’ responses, (2) differences between the model organism being used and human’s immune system, and (3) age-related variations in immune response.
Technology Overview
To address these challenges, Boston Children’s Hospital researchers have developed, MEMPHIS, a novel approach for identifying the ultimate formulation of effective vaccines through a platform that includes multiple in vitro analyses to provide a comprehensive understanding of underlying immunological responses at different levels.
This platform integrates four main modules: including analysis of leukocytes’ (white blood cells) responses for a specific individual, based on that individual’s specific immune system’s characteristics. Preliminary results demonstrated how one of the most effective adjuvant combinations (MPL and QS 21) activates multiple parts of the human immune system for different age groups, and how this comprehensive understanding of its mechanism can help in further development of vaccination adjuvants. As shown in the results readout summary below:
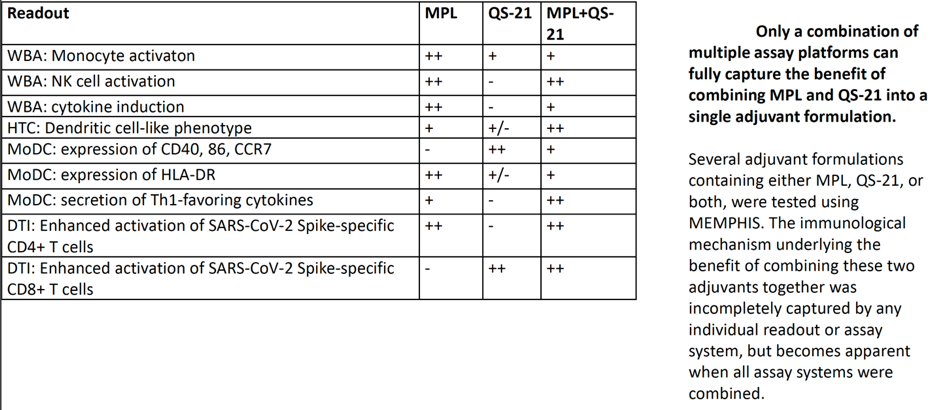
Applications
- Identifying the best adjuvant, or combination of adjuvants, to be used for a specific vaccine.
- Identification of the best adjuvant molecules to be used for vaccinating a specific age group of the population. For example, a vaccine used for neonates can utilize adjuvants that yield more efficient and longer-lasting immune response, in comparison to the same vaccine if it’s being administered to the adults’ age group.
- Through better and deeper understanding of the different mechanisms involved in different adjuvants and their combinations, it becomes possible to identify novel adjuvants, or combination of adjuvants, that provide better response to vaccination.
Benefits
- Reducing the number of individuals with failed vaccination.
- Providing life-long immunization for vaccines that used to require boosting over individuals’ lifetime.
- Increasing the efficacy and cost-effectiveness of vaccination during epidemics by providing more effective adjuvants to these vaccines.
- Reducing the adverse effects that prevent certain individuals from getting vaccines, including allergies to certain adjuvants, through the discovery of novel, safer, and more effective adjuvants, or combinations of these novel adjuvants.
Publications
- Oh, D.-Y. et al. Adjuvant-induced Human Monocyte Secretome Profiles Reveal Adjuvant- and Age-specific Protein Signatures. Mol Cell Proteomics 15, 1877–1894 (2016)
- Coccia, M. et al. Cellular and molecular synergy in AS01-adjuvanted vaccines results in an early IFNγ response promoting vaccine immunogenicity. Npj Vaccines 2, 25 (2017).
- Dowling, D. J. et al. TLR7/8 adjuvant overcomes newborn hyporesponsiveness to pneumococcal conjugate vaccine at birth. Jci Insight 2, e91020 (2017).