A rationally designed ICAM1 antibody drug conjugate for triple negative breast cancer and other ICAM1-expressing cancers
Background
Breast cancer is the most common cancer in women. It accounts for 12.5% of all cancers globally and women in the United States have a 1 in 8 chance of developing breast cancer. While the 5-year survival is about 90% for breast cancer, triple negative breast cancer (TNBC), which accounts for 15% to 20% of all breast cancers, has a 5-year survival rate of less than 77%.
Most TNBC cases are diagnosed at more advanced stages due to its aggressive nature and its limited ability to be detected through routine screening; in addition, being “triple-negative,” TNBC lacks the Estrogen-receptor (ER), Progesterone-receptor (PR), and the human epidermal growth factor receptor type 2 (HER-2), leading to poor responses to available therapies targeting these receptors. Other therapeutic modalities targeting specific molecular processes to cancer cells exist, but have limited efficacy, increased toxicity, and severe systemic adverse effects due to their limited specificity to cancer cells.
Technology Overview
In this invention, Dr. Marsha Moses and her team discovered a novel molecular target that is specific for TNBC cells, the intracellular adhesion molecule-1 (ICAM-1). Their extensive research demonstrated that linking chemotherapeutic agents to antibodies that are specific to ICAM-1 can make the cytotoxicity of these chemotherapies specific to TNBC cells, increasing the efficacy, safety, and success of chemotherapy for TNBC; a technique termed antibody drug conjugation (ADC).
The results below show the efficacy of using Antibody Drug Conjugate (ADC) of cytotoxic Dox therapy, in comparison to free Dox therapy, leading to tumor free mice following standard settings of drug administration:
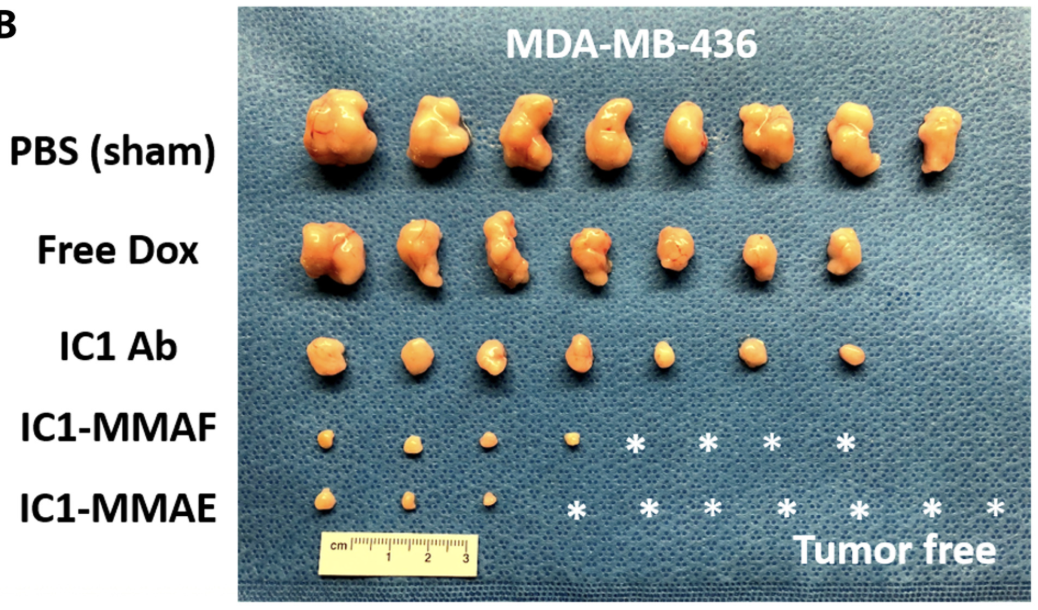
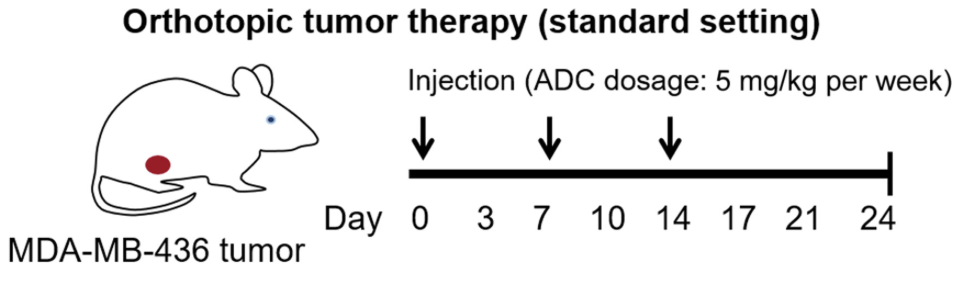
Benefits
- The ability to target, suppress and even eliminate triple negative breast cancer cells, for the first time, through a highly specific antibody-conjugated therapeutic agents.
- Reduced systemic toxicity and adverse effects of cytotoxic therapies, due to the high specificity for cancer cells.
- Increased cost-effectiveness of TNBC treatment, through increased efficacy of targeting the cancer cells, reduced need for repeated surgical resection, and minimal adverse effects of chemotherapy.
- The potential to utilize different chemotherapeutic agents to be antibody-conjugated, and the possibility of targeting other types of challenging cancer types that also express ICAM-1.
Applications
- Administration of ADC therapy for TNBC in earlier stages, to limit the possibility of metastasis and prevent the progression of cancer into advanced stages.
- Utilization of ADC as an adjuvant therapy following surgical resection, to eliminate micro metastases and increase the possibility of cancer remission.
- The potential to develop multiple antibody-conjugated cytotoxic therapies with ICAM-1 that can be combined with other therapeutic modalities, like immunomodulation, to increase the success rate of eliminating TNBC.